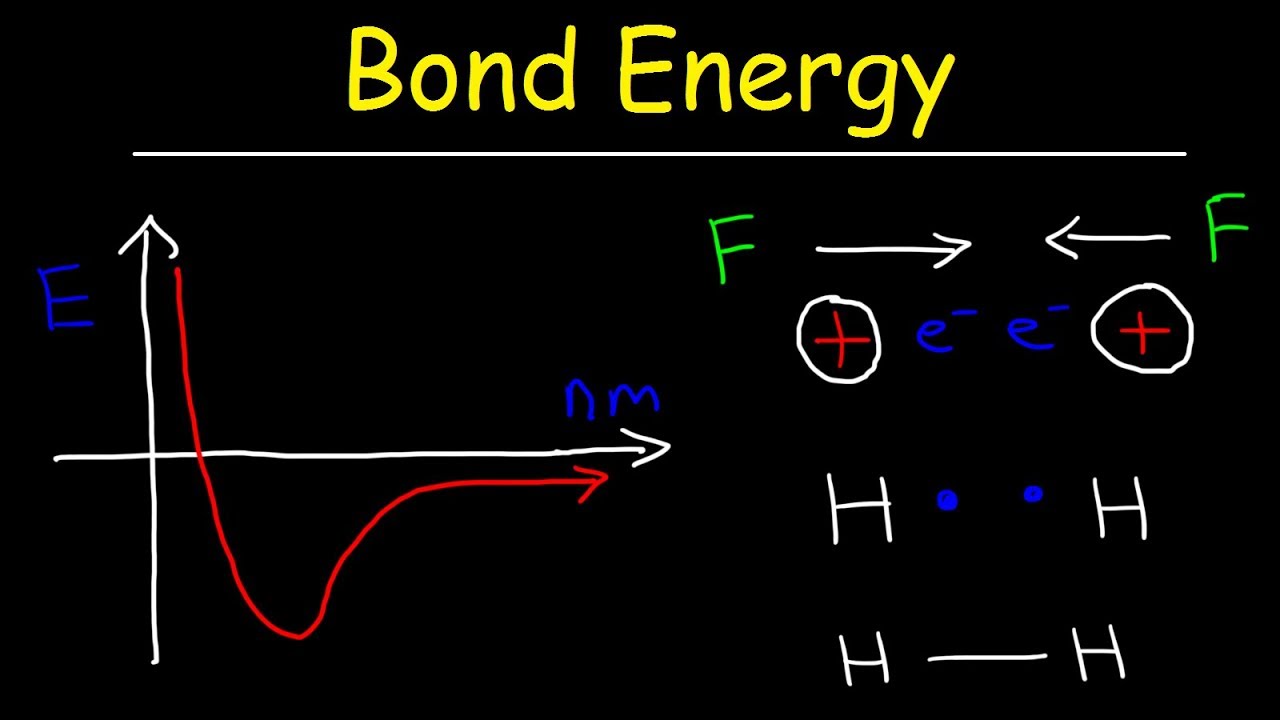
Bond Energy Diagram
Bond covalent energy potential bonding theory two lewis diagram atoms formation adichemistry between difference model when general 5.2: valence bond theory 89. chemical bonding (36)- covalent bonding(35) – molecular orbital
Potential Energy Diagrams For Formation Of Bonds | Mini Physics - Free
Chemistry bond energy potential chemical two covalent bonding atoms hydrogen electron versus between diagram valence ionic theory lewis structures water Bond energy potential distance atoms energies lengths two breaking molecule when length why covalent bonds curve formation between chemistry atom Bond energy strength 2021 helmenstine anne entry updated january posted may
9.8: second-row diatomic molecules
Enthalpy energies bonds calculations pptx teachingWhich of the following has highest bond energy? N2 bond order energy diagram level magnetic stability propertyTang 06 bond energy.
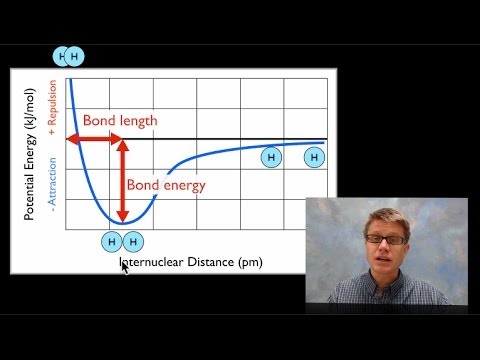
Bond length energy graph distance bondsBond chemistry energy bonding theory covalent valence distance atoms length two interaction system shown general graph hydrogen diagram between curve Energy bond exothermic diagram formation chemical bonds released when broken forming change endothermic negative enthalpy releases process always its reactionsBond order for o2.

Energy bond activation ppt powerpoint presentation bonds
9.8: second-row diatomic moleculesEnergy ion versus ionic bonding covalent chemical chemistry interactions bond distance when lattice system minimum basic potential interaction diagram internuclear (pdf) understanding the bond-energy, hardness, and adhesive force fromBond length and bond energy.
Bond energy chemical bonding formation length break required ppt powerpoint presentationIonic bonds Molecular orbitals diatomic orbital molecules valence electrons electron bonding paramagnetic delocalized libretexts principles heteronuclear chem pageindexBond energy.

Bond lengths assuming socratic dioxide methane chegg inorganic chemists quoted
5.4 valence bond theoryPotential energy diagrams for formation of bonds Hardness understanding electron bond adhesive phase function force energy diagram via workEnergy level diagrams, bonds energies, enthalpy..
O2 bond molecular moleculeEnergy potential bond diagram covalent formation waals der van bonds diagrams graph binding physics Potential energy diagrams for formation of bondsOrbital molecular bonding nitrogen theory molecule covalent chemical.

Bond energy and strength
Tang bonds chemical 01d reactionsBond lengths and energies Energy level diagram || bond order || magnetic property || stabilityBond energy and strength.
Bond energy length chemistry forces attraction repulsionChapter 4.1: ionic bonding Bond energy energies solutionBond energy & bond length, forces of attraction & repulsion.

Orbital molecules diatomic orbitals theory mo bonding of2 delocalized diagrams homonuclear electrons atomic chem libretexts lewis valence geometry correlation hybridization
Covalent bond .
.